 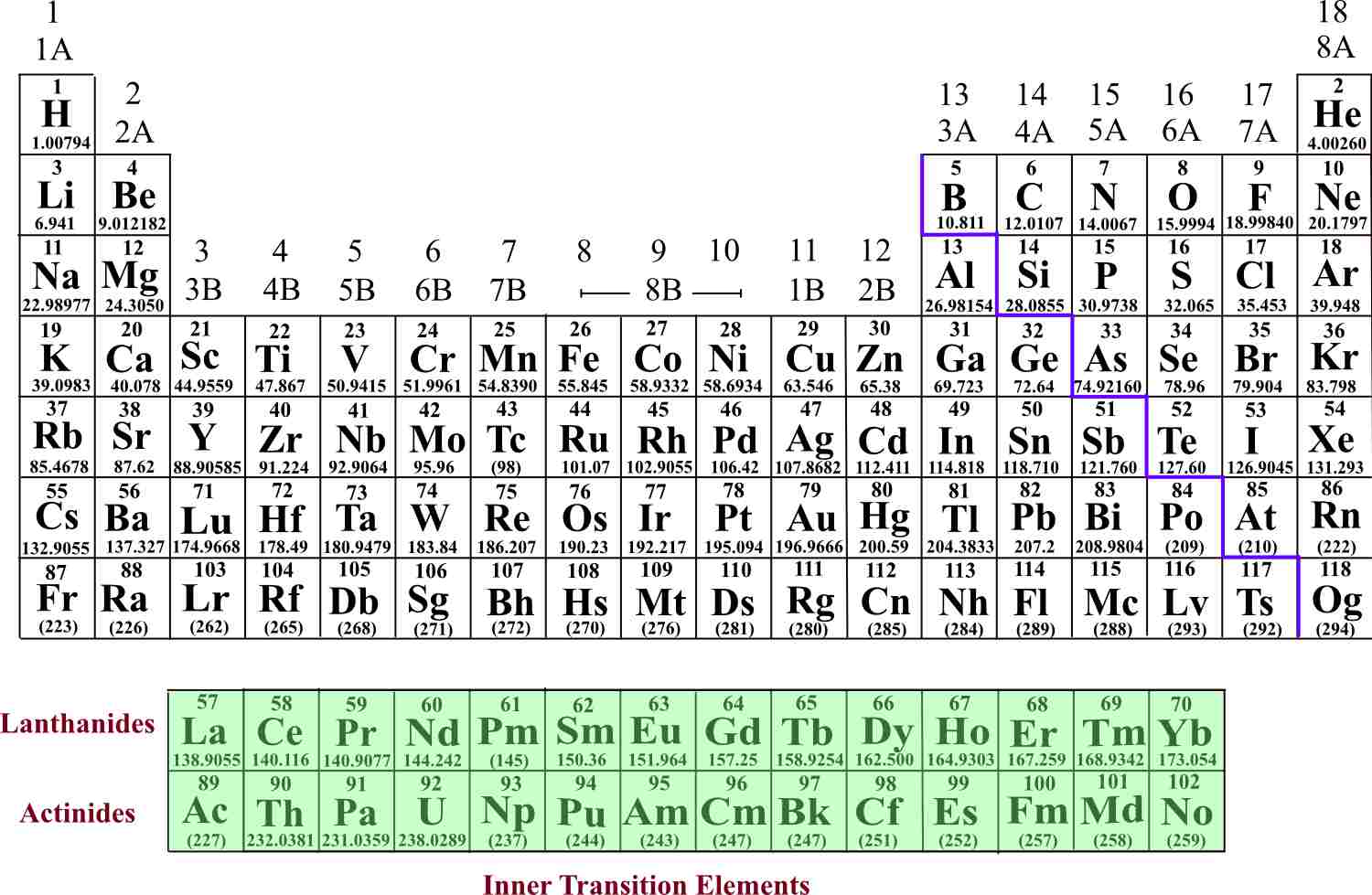
Thus transition metals tend to be very colorful, and each contributes different colors to different compounds. This configuration leads to a wide variety of available energy levels for electrons to move about in, so in the presence of electromagnetic radiation such as light, a variety of frequencies are readily emitted or absorbed. In most atoms, inner electron shells must be maximally occupied by electrons before an outer shell will accept electrons, but many transition elements have electron gaps in the shell just inside the valence shell. The rare-earth elements ( REE ), also called the rare-earth metals or rare earths or, in context, rare-earth oxides, and sometimes the lanthanides (although yttrium and scandium, which do not belong to this. The somewhat reactive elements that fall between the two extremes are the transition elements, such as iron, copper, tungsten, and silver. Refined rare-earth oxides are heavy, gritty powders usually brown or black, but can be lighter colors as shown here. (Hydrogen is exceptional in that, although it is highly reactive, its other metallic properties are manifest only at very high pressures.) Metals farther toward the right side of the Periodic Table, such as tin and lead, have more electrons in their outermost shell, and are not as reactive. Heavy metals (HMs) are defined as those elements having an atomic number greater than 20 and atomic density above 5 g cm 3 and must. Alkali metals, such as sodium and potassium, have only one electron in their outermost shell, and are chemically very reactive. With the exception of hydrogen, which behaves like a metal only at very high pressures, the elements that appear in the left-hand column of the Periodic Table are called alkali metals. Explainer: What is a metal A weak grasp on their electrons allows these elements to conduct electricity Molten iron, a metal, flows into a casting mold. In a substance composed of metals, the atoms are in a virtual sea of valence electrons that readily jump from atom to atom in the presence of an electric potential, creating electric current. The electrical conductivity of metals also stems from the relative freedom of valence electrons. For instance, most metals form ionic bonds easily because they readily give up valence electrons to other atoms, thereby becoming positive ions (cations). Many of their properties are due to the fact that their outermost electrons, called valence electrons, are not tightly bound to the nucleus. Most metallic elements are lustrous or colorful solids that are good conductors of heat and electricity, and readily form ionic bonds with other elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
